 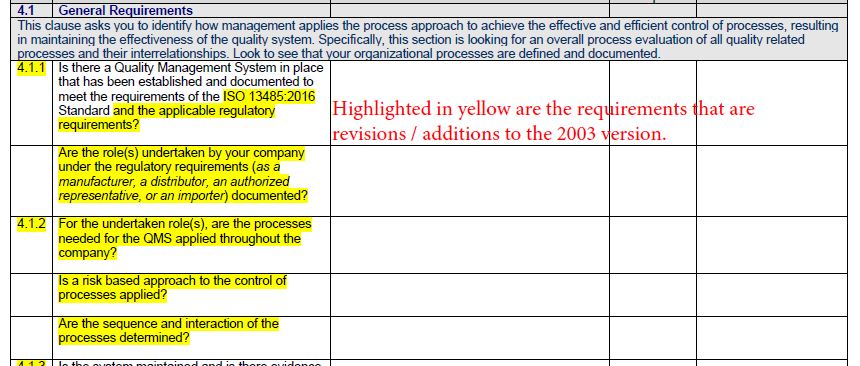
Essentially, it is a principle that all standards are structured the same way and can work The high-level structure consists of 10 clauses that push for more uniformity and integration among various One of the main changes introduced in the ISO 9001:2015 upgrade is the high-level Most likely include high-level structure and Annex L. It’s difficult to speculate what will be in the next version of the ISO 13485 Standard, but the update will What would an ISO 13485 revision look like? Risk management should not be a quality-only responsibility, it needs to be an all-hands endeavor integrated into allĪreas of the organization. Companies are encouraged to apply an iterative approach to With the standard’s risk management guidelines. As the sponsor organization, you must ensure that third-party organizations comply Standard also requires you to consider a risk-based approach for outsourced processes and suppliers.įor example, software suppliers are required to perform a risk-based assessment at every stage of the softwareĭevelopment life cycle (SDLC). The 2016 version requires a risk-based approach for theĮntire quality management system throughout the product’s life cycle - including design controls. 
Management principals in design controls. ISO 13485:2003 implies the need to incorporate risk 
What risk management concepts should I understand for ISO 13485:2016 compliance? Therefore, compliance with ISO 13485 standard will help with MDR compliance. The ISO 13485 standard is the medical device industry’s most widely used international standard for quality
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
